1,3-diaxial interactions
Investigate steric interactions in cyclohexane derivatives.
Tasks
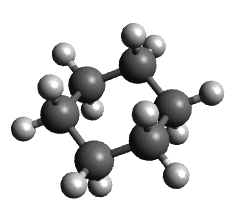
- Create and optimize a cyclohexane molecule
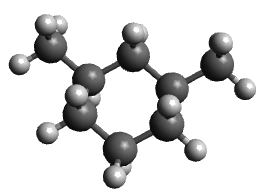
- Add methyl substituents to 1 and 3 positions in diequatorial positions, optimize and note the energy
- Delete diequatorial substituents, to return to cyclohexane, optimize again
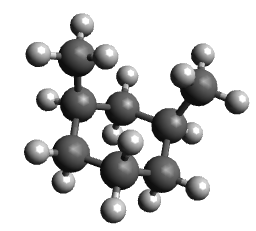
- Place methyl substituents in 1,3-diaxial positions, optimize and note the movement of the groups and the energy
Solution
 |
 |
 |
|
|---|---|---|---|
| Molecule | Unsubstituted cyclohexane | Diequatorial | Diaxial |
| Energy | 30 kJ/mol | 40 kJ/mol | 98 kJ/mol |

Comments